From pv magazine Global
Spanish scientist José Antonio G. I. has developed a system that is able to generate and store hydrogen on-site from tap water without electrolysis.
“Until now, the most common way to produce hydrogen is by electrolysis. However, this process implies a significant electrical consumption that makes it not interesting from an economic point of view,” he told pv magazine.
The prototype consists of a water tank that is initially filled with water and two more chemical elements that the researcher doesn’t want to reveal. Hydrogen production begins when a 20 W compressor discharges pressurized air in the lower part of the tank.
“It is the air that causes the reaction between the different chemical components and generates hydrogen,” the scientist explained. “Then, the generated hydrogen leaves through the upper area of the tank to be introduced into an equally pressurized tank. The water in this second tank collects the possible impurities and hydrogen comes out through the upper part of the tank.”
After the hydrogen is purified, it flows via an upper conduit to another tank equipped with closed contacts, a safety valve and an outlet pipe assisted by a solenoid valve.
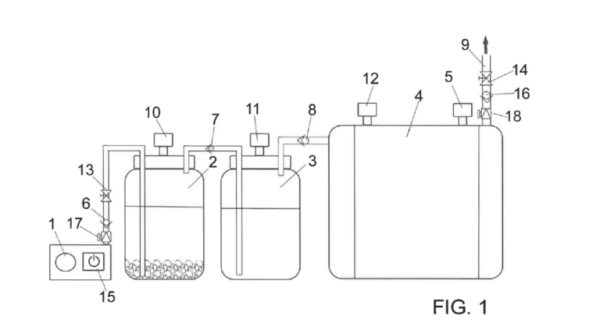
“Currently, we are developing a model with a 220-liter tank that can work with a pressure of 1 kg/cm2 and a flow rate of 30 liters per minute,” González Ibáñez went on to say. “This can generate heating, hot water and electricity for a household or a small business.”
The group is also designing a larger model that can work with a pressure of 750 Kg/cm2 and can reportedly supply a thermal power plant or a container ship.
“Hydrogen is produced at the place of consumption, so transportation disappears, it only needs a water tap,” the scientist emphasized. “The system is capable of generating the energy equivalent to a liter of gasoline –30 Mjoules or 8.333333 kW– for €0.0151515 ($0.0153749).”
Author:
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.







By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.